Rutherford atomic theory4/3/2024 
Mass number (A) of the atom is the total number of nucleons. The positive charge on the nucleus is due to protons, but the mass of the atom is due to protons and neutrons. = the number of electrons in a neutral atom Therefore, the number of electrons in sodium and hydrogen is 11 and 1, respectively.Ītomic number = the number of protons in the nucleus of an atom For example, the number of protons in sodium is 11 whereas it is 1 in hydrogen, Therefore, the atomic numbers of sodium and hydrogen are 11 and 1, respectively.Īlso, to maintain electrical neutrality, the number of electrons in an atom is equal to the number of protons (atomic number, Z). Atomic Number (Z) is the number of protons present in the nucleus. Also, the charge on the proton is equal but opposite to that of the electron. Atomic Number And Mass NumberĪs we know now, a positive charge on the nucleus is due to the protons. Now that we know the two atomic models, let’s try to understand a few concepts. 
Thus, Thomson and Rutherford’s atomic models revealed key aspects of the structure of the atom but failed to address some critical points. Rutherford’s model also does not state anything about the distribution of the electrons around the nucleus and the energies of these electrons.Then the electrostatic attraction between the electrons and the dense nucleus will pull the electrons into the nucleus to form a miniature version of Thomson’s model. Contrarily, let’s consider that the electrons do not move and are stationary.Thus, Rutherford’s model does not explain the stability of the atom. If this is true, then the electron will spiral into the nucleus. Therefore, an electron in an orbit will emit radiation and eventually the orbit will shrink. According to Maxwell’s electromagnetic theory, charged particles when accelerated must emit electromagnetic radiation. Since a body that moves in an orbit must undergo acceleration, the electrons, in this case, must be under acceleration. According to Rutherford’s atomic model, the electrons (planets) move around the nucleus (sun) in well-defined orbits.Electrostatic forces of attraction hold the nucleus and electrons together.Therefore, it is also referred to as the Planetary Model. This arrangement also resembles the solar system, where the nucleus forms the sun and the electrons are the revolving planets. Electrons surround the nucleus and move around it at very high speeds in circular paths called orbits.Rutherford called this region the nucleus. Most of the mass of the atom and the positive charge is densely concentrated in a very small region in the atom.Rutherford’s calculations show that the volume of the nucleus is very small compared to the total volume of the atom and the radius of an atom is about 10 -10m, while that of the nucleus is 10 -15m.īased on his observations and conclusions, Rutherford proposed his model of the structure of the atom.The positive charge has to be concentrated in a very small volume to deflect the positively charged α-particles. This suggests that the positive charge is not uniformly spread throughout the atom as Thomson had proposed. The deflection of a few positively charged α-particles must be due to the enormous repulsive force.Since most of the α-particles passed through the foil undeflected, most of the space in the atom is empty.Thomson’s model versus Rutherford’s model Conclusions of the α-scattering experimentīased on the above results, Rutherford made the following conclusions about the structure of the atom: A tiny flash of light was produced at a point on the screen whenever α-particles struck it. A circular, fluorescent zinc sulfide screen was present around the thin gold foil. In this experiment, high energy α-particles from a radioactive source were directed at a thin foil (about 100nm thickness) of gold. α-Particle Scattering Experiment Experiment To come up with their model, Rutherford and his students – Hans Geiger and Ernest Marsden performed an experiment where they bombarded very thin gold foil with α-particles. 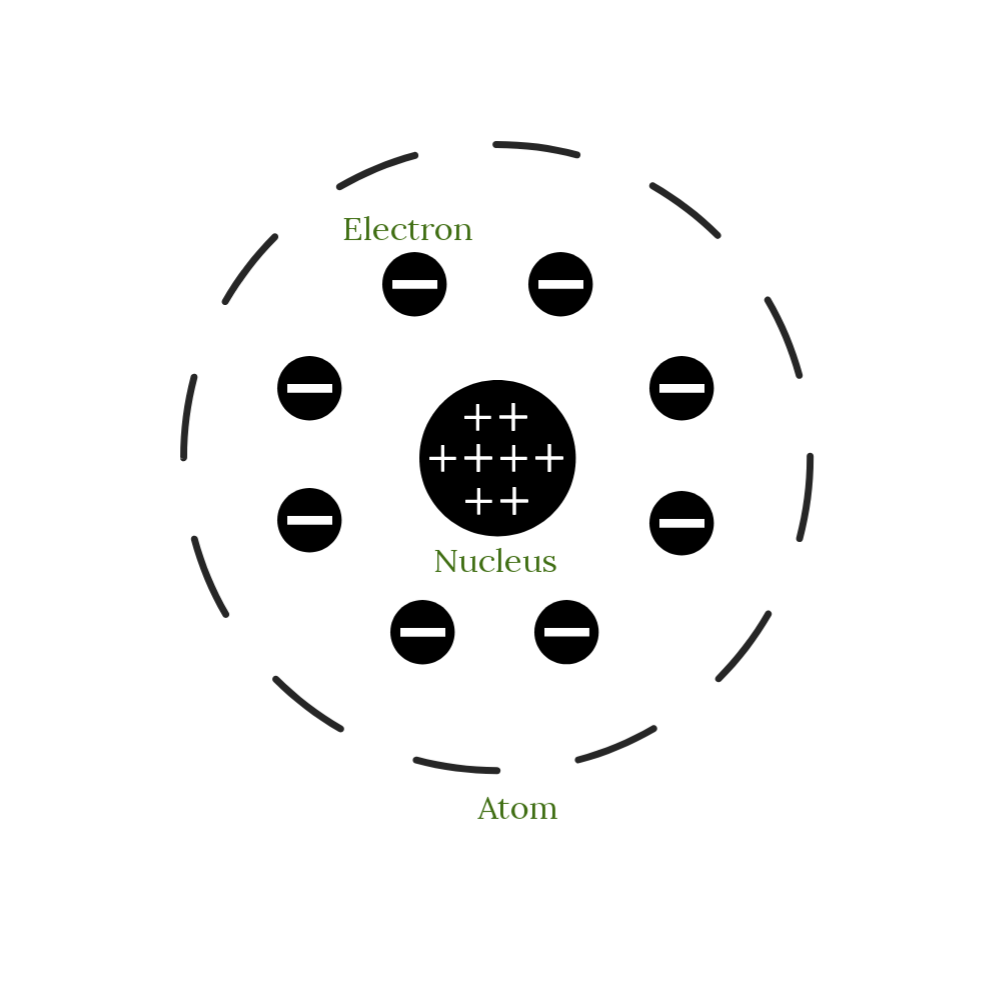
The second of the atomic models was the contribution of Ernest Rutherford. You can download Structure of Atom Cheat Sheet by clicking on the download button below Thomson was awarded the Nobel Prize in physics for his theories and experiments on electricity conduction by gases. However, its propositions were not consistent with the results of later experiments. Thomson’s atomic model was successful in explaining the overall neutrality of the atom. 
Therefore, this model is also referred to as the watermelon model, the plum pudding model or the raisin pudding model.Īn important aspect of this model is that it assumes that the mass of the atom is uniformly distributed over the atom. Thomsons’ atomic model Doesn’t the figure above remind you of a cut watermelon with seeds inside? Or, you can also think of it as a pudding with the electrons being the plum or the raisins in the pudding.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
